AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Density of water si4/11/2024   /experiment-showing-relative-density-of-different-liquids-liquids-of-different-densities-will-sit-on-top-of-each-other-without-mixing-and-if-mixed-together-will-re-settle-into-layers-the-most-dense-liquid-lies-at-the-bottom-the-least-dense-at-the-t-57a768ac3df78cf459161869.jpg)
Bulk modulus for water - Imperial Unitsĭensity of a fluid changing both Temperature and Pressure Note! Bulk modulus for liquids varies with pressure and temperature. Ρ 1 = ρ 0 / (1 - (p 1 - p 0 ) / E) (6b) Bulk Modulus Fluid Elasticity some common Fluids - E or combined with (2) - the final density can be expressed as With (5) - the final volume after pressure change can be expressed asĬombining (5b) with (1) - the final density can be expressed as: The minus sign corresponds to the fact that an increase in the pressure leads to a decrease in volume. = - (p 1 - p 0 ) / ((V 1 - V 0 ) / V 0 ) (5)Į = bulk modulus - liquid elasticity (N/m 2) The influence of pressure on the volume of a liquid can be expressed with the three dimensional Hooke's law Note! - volumetric temperature coefficients may vary strongly with temperature. volumetric expansion coefficient for some commonly used materials.Ρ 0 = initial density (kg/m 3 ) Volumetric Temperature Coefficients - β With (1) and (3b) the final density after a temperature change can be expressed as Β = volumetric temperature expansion coefficient (m 3 /m 3 o C)ĭt = t 1 - t 0 = change in temperature - difference between final and initial temperature ( oC) When temperature increases - most liquids expands:ĭV = V 1 - V 0 = change in volume - difference between final and initial volume (m 3 ) V = specific volume (m 3 /kg) Volume and change in Temperature The inverse of density is specific volume: The density of a liquid can be expressed as See also Water - Density, Specific Weight and Thermal Expantion Coefficient, for online calculator, figures and tables showing changes with temperature. The density of water versus temperature and pressure is indicated below: Remember that the density of water may change under extreme conditions, but if you are measuring it around room temperature with minimal impurities present, you can rely on this method for accurate results.The density of a liquid will change with temperature and pressure. Understanding and measuring the density of different substances, including water, plays a crucial role in many real-life applications such as designing ships or studying ocean currents. So, the density of water at room temperature is approximately 1,000 kg/m³.īy following these simple steps, you can determine the density of water at room temperature. 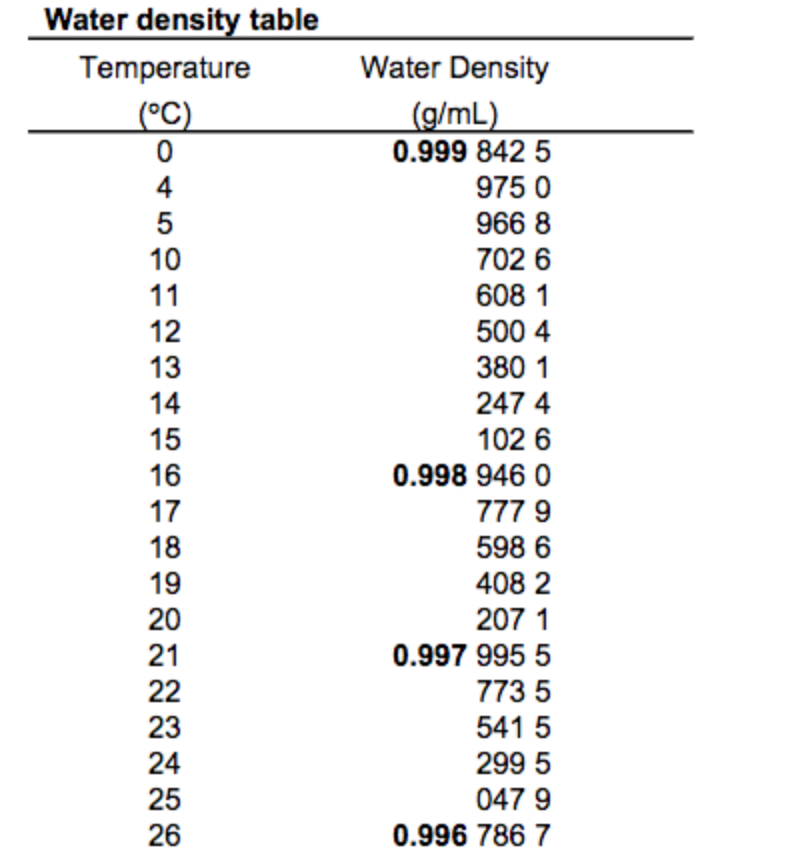
Now, we apply the formula to calculate the density: We weigh our filled container and find out that the mass is 500 g (0.5 kg). Let’s consider a sample of 500 mL (0.5 L) of water taken at room temperature. Now that you have both mass and volume values for your water sample, you can proceed to calculate its density using the formula mentioned above. Then pour your measured water sample into the container and record its mass (m). Place an empty container on the scale and tare (zero) it. You can use either a graduated cylinder or a volumetric flask for more accurate results. I) A container or a beaker, preferably with volume markingsįill the container or beaker with a known volume of water (V). To calculate the density of water, you will need: The standard SI units for density are kilograms per cubic meter (kg/m³). To determine the density of water, we need to measure its mass and divide it by its volume. 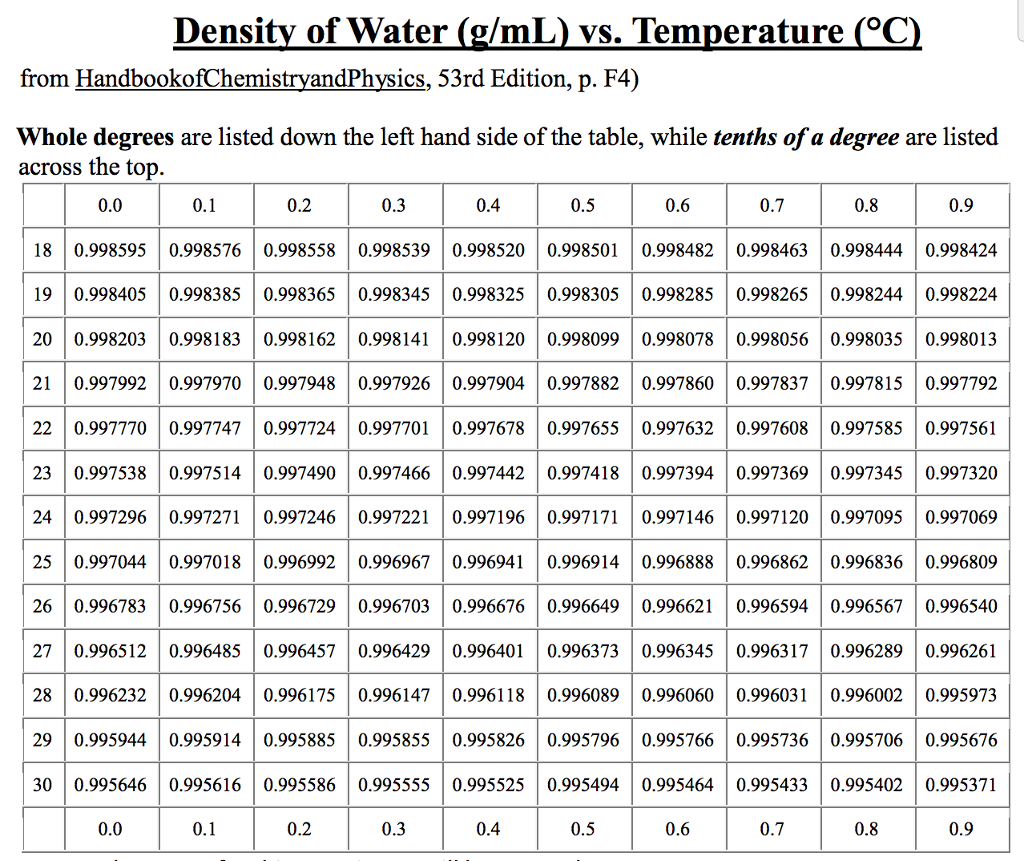
But, for simplicity purposes, we will look at the method for calculating the density of pure water at room temperature.ĭensity (ρ) can be calculated using the following formula: Water’s density varies based on factors such as temperature and impurities. In this article, we will discuss how to calculate the density of water. It describes the mass of an object per unit volume, giving us valuable information about the substance’s behavior and characteristics. Density is an important concept in various fields like physics, chemistry, and engineering. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
